Our Approach
Bariatric surgery (e.g., “Roux-En-Y bypass) now called metabolic surgery, performed for weight loss, shifts digestion of food from the upper small intestine to the lower part of small intestine and consequently improves type 1 diabetes and resolves type 2 diabetes within days in a majority of patients with diabetes. In addition, lowering of cholesterol, blood pressure, triglycerides, and C-reactive protein has been observed in these patients prior to significant weight loss (Buchwald, H. et.al; JAMA, 2004; 292:1724-1737). These effects have led to bariatric surgery being commonly referred to as bariatric-metabolic surgery. There is strong evidence that the mechanism of this effect results from expedited delivery of specific nutrients to the lower gut that promote secretion of gut hormones crucial for the regulation of blood glucose and lipids. BioKier’s approach is to deliver an active nutrient to the lower gut via a tablet, thus mimicking bariatric-metabolic surgery and promoting secretion of gut hormones with the need for surgery.
BioKier is a clinical-stage company developing oral products for the management of glucose and lipids that can be marketed as prescription drugs and nutritional supplements. These products are designed to mimic bariatric-metabolic surgery which is known to resolve diabetes and improve cardiometabolic health. Completed clinical trials show that BioKier’s approach improves gut hormone secretion and regulation of glucose and lipids.
The following animation shows:
- Nutrients in food stimulate hormone responses in non-diabetic subjects
- Diabetes affects this response
- RYGB corrects the effects of diabetes
- Targeted delivery of BioKier’s capsule will mimic the effects of RYGB
- Progress in product development and initial clinical results
- New grants and upcoming clinical trials
BioKier has devised two proprietary oral formulations, one a tablet containing butyrate (BKR-017) and the other a capsule containing glutamine (BKR-013), that are designed to mimic the effects of metabolic surgery and improve glucose management without the physical trauma and cost of the surgery. BioKier’s approach is expected to be suitable for type 1 and type 2 diabetes patients, both obese and non-obese. Specifically, both products act by stimulating L-cells in the lower gut thus correcting the deficiency in secretion of endogenous GLP-1 and other gut hormones, such as PYY, GLP-2, and oxyntomodulin, that regulate glucose metabolism and appetite. Therefore, this treatment also is expected to have beneficial effects on hypertriglyceridemia, metabolic syndrome, and obesity.
BioKier has devised several proprietary oral formulations with two active ingredients:
Butyrate Tablets:
BKR-017 (sustained-release)
BKR-021 (rapid-release)
Glutamine Capsule:
BKR-013 (rapid-release)
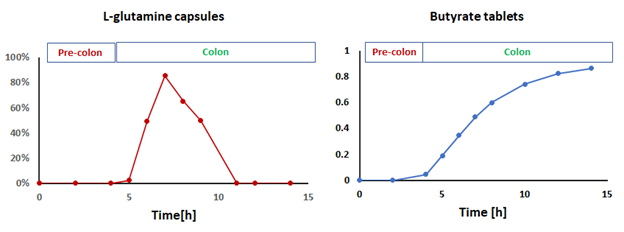
All of these products are designed to mimic the effects of metabolic surgery and improve cardiometabolic health conditions such as hyperlipidemia (predominantly cholesterol), diabetes, and fatty liver without the physical trauma and cost of the surgery. BioKier’s approach is expected to be suitable for patients with high lipids, type 1 or type 2 diabetes, and fatty liver, both obese and non-obese. Specifically, the products act by stimulating L-cells in the lower gut, thus correcting the deficiency in secretion of endogenous gut hormones which is observed in diabetes, insulin resistance, and obesity. The gut hormones, which include GLP-1, PYY, GLP-2, and oxyntomodulin, regulate glucose metabolism, lipid levels, and appetite. Therefore, this treatment also is expected to have beneficial effects on hypertriglyceridemia, metabolic syndrome, and obesity.

